In Vitro Evaluation of Oil Seed Cakes (Oscs) as Sustainable Soil Amendments for the Management Sclerotium Rolfsii Pathogen in Groundnut
0 Views
R. MANASA*, R. SARADA JAYALAKSHMI DEVI, HARI KISHAN SUDINI, VEMANA, K. JOHN AND G. RAMA RAO
International Crops Research Institute for the Semi-Arid Tropics, Patancheru 502324.
ABSTRACT
The study evaluated the antifungal activity of six oil seed cakes (OSCs) namely neem, groundnut, sesame, mustard, castor and a mixture cake against Sclerotium rolfsii using a divided plate assay and poison food technique. Sesame cake demonstrated the highest efficacy, achieving 56.10 per cent mycelial inhibition with unautoclaved sesame cake, compared to 14.63 per cent with autoclaved cake. In aqueous extract assays, sesame cake achieved complete inhibition (100%) of fungal growth at a 25 per cent concentration, with significant inhibition observed at lower concentrations (93.80% at 20%, 71.98% at 15%, and 50.91% at 10%). Additionally, sesame cake reduced the formation of sclerotial bodies, with counts dropping to 15 at a 15 per cent concentration. In contrast, castor and neem cakes exhibited no inhibitory activity at any concentration, while mustard and mixture cakes displayed moderate antifungal effects, with maximum inhibition rates of 28.95 per cent and 31.72 per cent, respectively.
The antifungal efficacy of sesame cake was attributed to its bioactive constituents, as identified through Gas Chromatography-Mass Spectrometry (GC-MS) analysis. Key bioactive compounds included oleic acid (42.62%), linoleic acid derivatives (31.58%), palmitic acid (13.86%), and sesamol (1.34%), which are known for their antimicrobial properties. At 3.0 per cent concentration, sesame cake reduced mycelial growth to 1.67 cm, corresponding to a 79.17 per cent inhibition rate. The findings of the study highlight sesame cake as a potent antifungal agent against S. rolfsii, with its bioactive components acting synergistically to inhibit fungal growth and reduce sclerotial formation, supporting its potential as a sustainable soil amendment in the management of soil borne pathogens.
KEYWORDS: Oil seed cakes, S. rolfsii, in vitro conditions, antifungal activity and GC MS analysis.
INTRODUCTION
Groundnut (Arachis hypogaea L.), an allotetraploid crop (2n=4x=40, AABB genome), is a vital oil seed crop cultivated across tropical and subtropical regions of the world (Bosamia et al., 2020). Known by various names like peanut, poor man’s cashew nut, earthnut and monkey nut, it is believed to have originated in South America, particularly in Bolivia, Paraguay, Peru, Northern Argentina and Western Brazil. Today, groundnut is grown in over 100 countries, covering 327 lakh hectares globally, with a production of 539 lakh tonnes and an average productivity of 1648 kg/ha. India leads in cultivation area and ranks second in production, contributing 101.35 lakh tonnes with a productivity of 1777 kg/ha (FAOSTAT, 2021). However, India’s productivity is lower than that of the USA (4497 kg/ha) and China (3913 kg/ha) (USDA, 2023). Major groundnut-growing states in India, including Gujarat, Andhra Pradesh, Rajasthan, Karnataka, Madhya Pradesh, Tamil Nadu and Maharashtra, contribute 80% of the country’s production. According to estimates, production reached 11.90 million tons from 5.75 million ha with a average yield of 2067 kg/ha in 2024-24 (Annual report, 2024).
Groundnut production faces numerous challenges due to poor soil fertility and various biotic and abiotic stresses (Pujer et al., 2021). Among biotic constraints, Sclerotium rolfsii is a significant soil-borne fungal pathogen causing stem rot and pod rot. The disease, first reported by Mc Clintock (1917) in Virginia, USA, is also known as white mold or southern blight. Stem rot leads to yield losses ranging from 10 per cent to 40 per cent, but severe outbreaks can reduce yields by upto 80% (Bera et al., 2014). The pathogen affects pegs and pods, causing substantial harvest losses. In heavier soils, the infection is usually limited to the soil surface, while in lighter soils, it can penetrate deeper, affecting pods and pegs. Moist and warm weather favors stem rot development, while drier soil conditions promote pod rot (Mehan and Mc Donald, 1990).
Managing stem rot is difficult due to the absence of resistant groundnut varieties and the pathogen’s persistence in the soil. Fungicide application offers partial control but increases production costs and poses environmental hazards such as soil infertility, pollution, and contamination of water bodies. Continuous fungicide use may also lead to pathogen resistance (Mehan et al., 1995). Therefore, sustainable, non-chemical approaches to disease management are essential. An integrated strategy combining host resistance, cultural practices, and biological control is necessary to manage stem rot effectively. Organic amendments, particularly oil seed cakes (OSCs), have demonstrated efficacy in suppressing soil-borne diseases due to their soil-enhancing properties (Adiver, 2003). These amendments provide essential nutrients to crops and stimulate the activity of beneficial bioagents. Neem cake, for example, increases soil nitrate availability, improving nitrogen metabolism in plants. The use of organic amendments like green manures, farmyard manure, compost and oil cakes has been linked to enhanced plant health and reduced disease incidence (Sivaprakasam, 1991). Blum and Rodriguez-Kabana (2004) highlighted the dual role of organic amendments in promoting plant growth and controlling soil-borne pathogens. Their ability to improve microbial activity in soil contributes to natural disease suppression.
Oil seed cakes such as neem, groundnut, sesame, mustard, castor, and mixed cakes exhibit suppressive effects against soil-borne pathogens by releasing bioactive compounds that inhibit fungal growth. These amendments have been shown to reduce the growth and sclerotia formation of S. rolfsii. Their integration into disease management strategies helps reduce the reliance on chemical fungicides. Blum and Rodriguez-Kabana (2004) emphasized the importance of organic inputs in improving soil health and enhancing resistance to soil-borne diseases. The use of OSCs as part of an integrated management strategy can significantly lower the environmental impact of disease control and support sustainable production practices for groundnut farmers. The present study aimed to evaluate the inhibitory potential of oil seed cakes (OSCs) against S. rolfsii under in vitro conditions.
MATERIAL AND METHODS
i. Source of the pathogen
The pathogen was isolated using two methods:
A. Direct isolation
Under aseptic conditions, a sterile pointed needle was used to pick fungal growth or sclerotia directly from infected groundnut stems. The collected material was transferred onto plates containing Potato Dextrose Agar (PDA). The plates were then incubated at 26 ± 2°C to allow fungal growth. Sclerotial bodies were collected from 14 days old culture plates and subsequently stored at 4°C for use in further studies.
B. Tissue isolation method
Infected stem segments were first washed thoroughly with tap water and then cut into small pieces. These pieces were surface sterilized using 1% NaOCl for 1 min, followed by three rinses with sterile distilled water under aseptic conditions. The sterilized tissue pieces were placed on PDA plates inside a laminar airflow chamber. The plates were incubated in a BOD incubator at 26 ± 2°C to promote fungal growth.
Hyphae emerging from the infected tissue were carefully sub-cultured onto fresh PDA plates. Pure cultures were obtained using the hyphal tip technique and further purified using a single sclerotium. The purified culture was examined microscopically for identification and maintained on PDA for subsequent studies. The initial isolation of the pathogen was carried out from groundnut plants exhibiting characteristic stem rot symptoms in the Patancheru fields.
ii. In vitro evaluation of aqueous extracts of OSCs against rolfsii
To prepare aqueous extracts of OSCs, 100 g of each OSC was powdered and soaked in sterile distilled water at a ratio of 1 g to 1.25 mL of water. The mixture was left to stand overnight, after which it was ground further using a pestle and mortar. The resulting material was filtered through a double layer of muslin cloth, and the filtrate was centrifuged at 10,000 rpm for 15 min. The supernatant was used as the standard organic amendment extract (100%) (Dubey, 2002 and Jangir et al. 2020). Before incorporation into the medium, the extract was sterilized at 1.4 kg cm⁻² pressure for 15 min. A specific volume of the sterilized extract was aseptically mixed

with molten PDA to achieve the desired concentration.
To prevent bacterial contamination, a small amount of streptomycin sulphate (0.5g/1L) was added to the medium prior to pouring it into sterilized Petri plates. Each plate contained approximately 20 mL of the prepared medium, which was allowed to solidify. A 5 mm disc of 7 days old S. rolfsii culture was placed at the center of each plate. Plates without the extract served as the control. Each treatment, along with the control, was replicated three times. The inoculated plates were incubated at 28±2°C in a BOD incubator. Mycelial growth was monitored and sclerotia were counted 15 days post-inoculation (Thakkar et al., 2018). The percent inhibition (PI) of mycelial growth was calculated using the following formula:
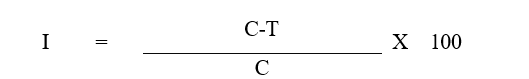
Where:
I = Percent inhibition
C = Radial growth (cm) in control
T = Radial growth (cm) in treatment
- In vitro assay for volatile compounds (VOCs) against rolfsii
The inhibitory effects of volatile compounds (VOCs) produced by OSCs were assessed using a divided plate assay. Powdered OSCs (5g, dry weight) were placed in one compartment of the divided plate, while PDA was poured into the other half and inoculated with a 5 mm mycelial disc of 7 days old S. rolfsii culture. Control plates consisted of PDA with only the fungal disc. Mycelial growth of the pathogen was recorded, and PI was calculated using the previously mentioned formula (Jangir et al., 2020).
iii. Extraction and identification of antifungal bioactive compounds from OSCs (GC-MS analysis)
Bioactive compounds were extracted from aqueous OSC extracts using hexane, a low-polarity solvent. A 1:1 ratio of aqueous extract to hexane was prepared and shaken overnight to ensure complete extraction of compounds. The mixture was centrifuged at 10,000 rpm for 10 minutes, and the supernatant was collected. The hexane was removed using a rotary evaporator. The resulting extract was dissolved in DMSO (Dimethyl Sulphoxide – a neutral solvent) and filtered through a 0.45 μm Millipore filter. The filtrate was then tested for antifungal activity using the poisoned food technique. PDA medium was poisoned with various concentrations of the metabolite (0.1%, 0.5%, 1%, 1.5%, 2%, 2.5%, and 3%), and the medium was poured into sterilized petri plates. Once solidified, a 5 mm disc of S. rolfsii was placed at the center of each plate. Control plates contained PDA poisoned with only DMSO. All plates were incubated at 28°C for 7 days. Mycelial growth was recorded, and PI was calculated using above mentioned formula. The most effective extract was subjected to gas chromatography-mass spectrometry (GC-MS) analysis for compound identification (Jangir et al., 2020).
For GC-MS analysis, a Shimadzu gas chromatograph equipped with a Turbo Mass Gold detector was used. The system included an Elite-1 column (100% Dimethyl Poly Siloxane) measuring 30 m × 0.25 mm ID × 1 mm df. The conditions for analysis were as follows: helium was used as the carrier gas at 1 mL/min; the oven temperature was programmed from 110°C (2 min hold) to 280°C (9 min hold); the injector temperature was set at 250°C, with a total GC run time of 45 minutes. A 1 mL aliquot of the hexane extract was injected into the system. The constituents were identified using a computer-assisted algorithm that matched the mass spectrum to the National Institute of Standards and Technology (NIST) library (Version 2.0, 2005). The GC-MS analysis was performed using Turbo Mass 5.1 software. This work was conducted in the Department of Entomology, ACI Programme, ICRISAT, Patancheru, Hyderabad.
RESULTS AND DISCUSSION
In recent years, the utilization of sustainable soil amendments, particularly oilseed cakes, has garnered significant attention for their potential role in managing soil borne diseases, including stem rot caused by S. rolfsii in groundnut. The persistence of the pathogen, attributed to its production of long-lived sclerotia, presents a formidable challenge to conventional chemical control strategies, necessitating the exploration of environmentally sustainable alternatives. Oilseed cakes, such as neem and castor cakes, are known for their high nitrogen content and antifungal properties, which enable them to suppress soil borne pathogens while simultaneously enhancing soil fertility. Additionally, their capacity for slow nutrient release supports long-term soil health and microbial activity. This study aims to evaluate
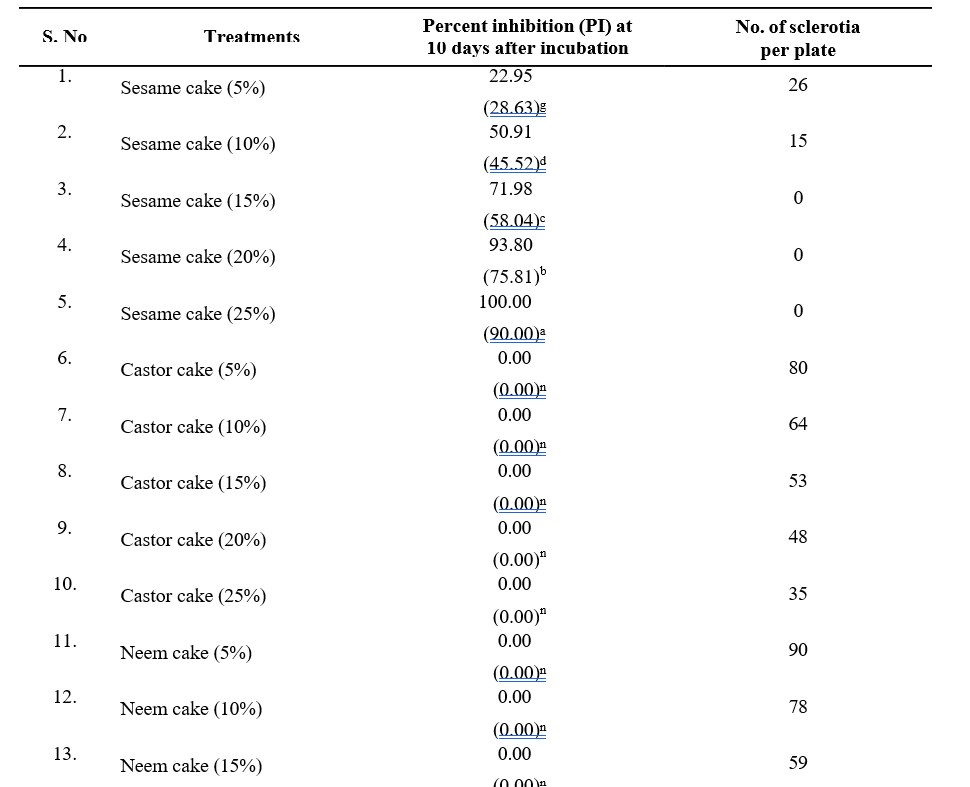
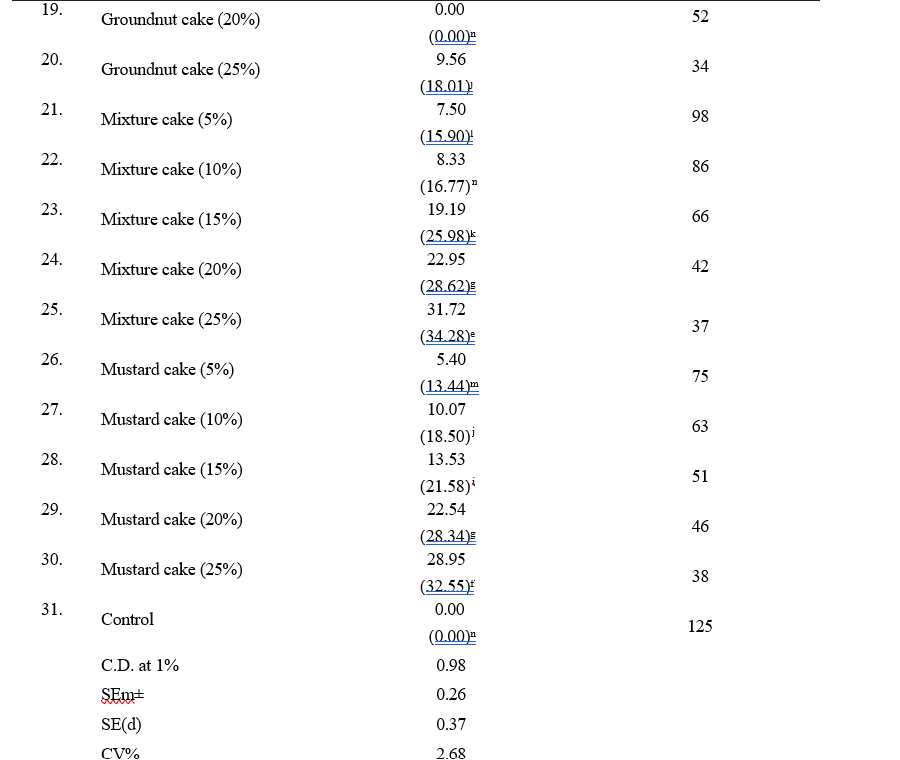
Table 1. In vitro evaluation of aqueous extracts of OSCs against S. rolfsii

*Values are the means of three replications in the table
Figures in parentheses represent angular transformed values. Means in a column followed by same superscript letters are significantly different according to DMRT test.
Note: C.D., critical difference; SE(d), standard error mean difference; CV, coefficient of variation.
the efficacy of different oilseed cakes in mitigating stem rot incidence, thereby contributing to the development of Integrated Disease Management (IDM) practices that align with sustainable agriculture objectives.
i. Antifungal activity of OSCs in volatile assay
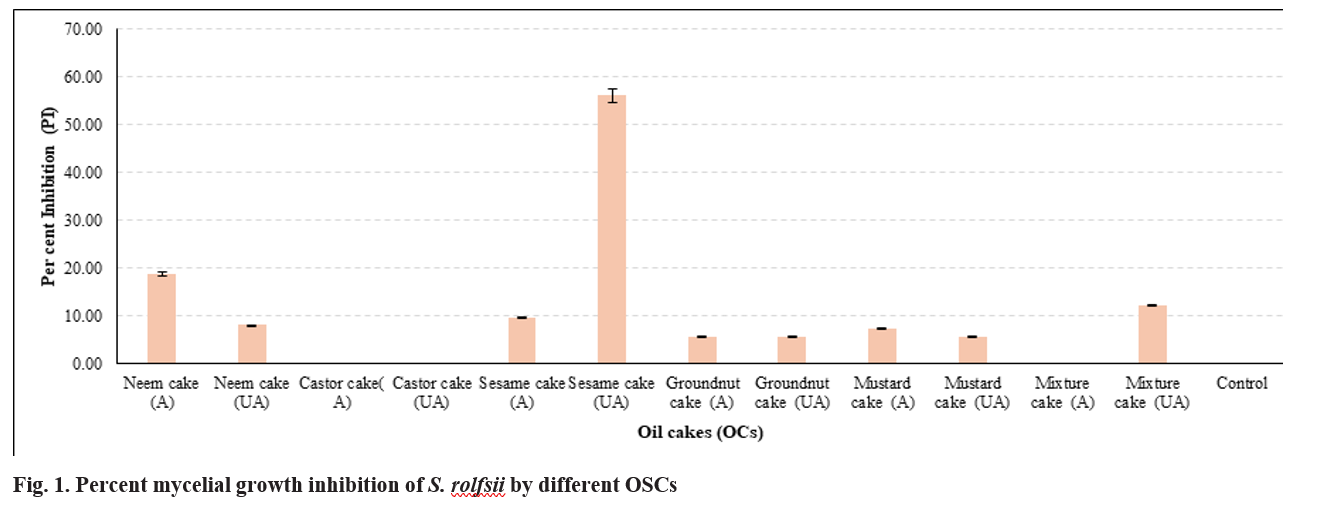
A total of six OSCs, including neem cake, groundnut cake, sesame cake, mustard cake, castor cake and a mixture cake (groundnut, flax seed and cotton seed), were evaluated for their antifungal efficacy against S. rolfsii. The inhibitory potential of volatile compounds (VCs) produced by these cakes was assessed using a divided plate assay. Among the tested OSCs, sesame cake exhibited the highest inhibition of mycelial growth, as evidenced by the reduced mycelial diameter of S. rolfsii, which measured 1.80 cm in the presence of unautoclaved sesame cake compared to 3.70 cm with autoclaved sesame cake. This corresponded to inhibition rates of 56.10 per cent and 14.63 per cent, respectively (Fig. 1&2). Additionally, the unautoclaved neem and mixture cakes exhibited inhibition rates of 18.70 per cent and 12.20 pe cent, respectively.
The superior inhibitory potential of sesame cake observed in the study is consistent with the findings of Jangir et al. (2020), who reported significant inhibition of Fusarium oxysporum using OSCs. Mustard cake was found to be the most effective in their study, achieving inhibition rates of 69.93 per cent (unautoclaved) and 40.51 per cent (autoclaved). Although the fungal pathogens differ, the inhibitory activity of OSCs against fungal growth is evident, suggesting that the response of fungal pathogens to OSC-derived volatile compounds may be species-specific. The use of a divided plate assay in both studies underscores the effectiveness of volatile compounds in inhibiting fungal mycelial growth. A comparative analysis of neem cake in the study and that of Jangir et al. (2020) revealed differences in inhibitory potential. In the study, unautoclaved neem cake inhibited S. rolfsii by 18.70%, while Jangir et al. (2020) reported a higher inhibition rate of 34.96 per cent against F. oxysporum. The variation could be attributed to differences in pathogen biology, the composition of volatile compounds, or experimental conditions. Collectively, the studies demonstrate the capacity of OSCs to inhibit soilborne fungal pathogens, though their efficacy is influenced by the pathogen type and OSC composition.
Further, the antifungal potential of volatile compounds was validated by the study carried by Thangaraj et al. (2023), who investigated the antifungal activity of volatile compounds emitted by medicinal herbs against Pythium aphanidermatum using the sealed plate method. Their results showed that volatiles from Mentha spicata were the most effective, achieving 45.56 per cent inhibition, followed by Cymbopogon citratus (24.70%) and Vitex negundo (18.88%). The finding aligns with the present study’s observation of high inhibition rates associated with sesame cake volatiles, highlighting the potential of plant-based volatile compounds for controlling soilborne pathogens.
ii. Antifungal activity of aqueous extract of oil seed cakes (OSCs)
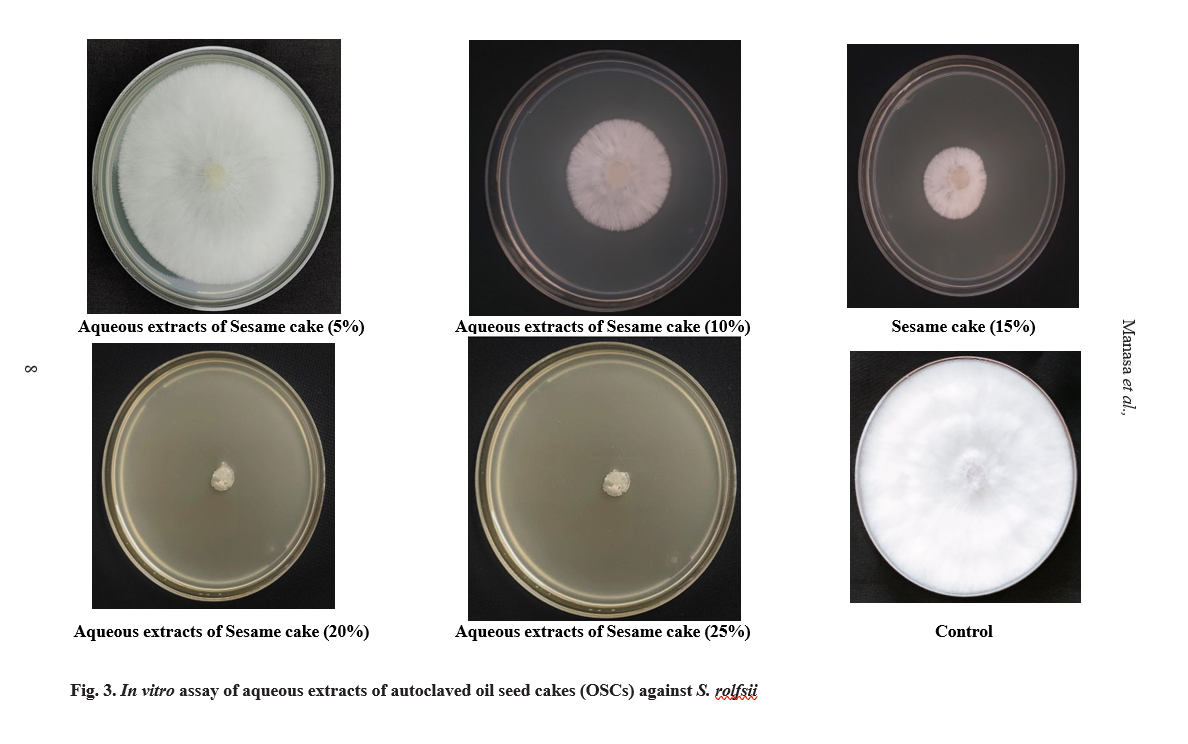
The antifungal efficacy of aqueous extracts of the six OSCs was evaluated against S. rolfsii at concentrations of 5, 10, 15, 20, and 25% using the poisoned food technique. The PI of mycelial growth and the number of sclerotial bodies produced after 10 days of incubation were recorded. Sesame cake demonstrated the highest inhibitory potential, achieving complete inhibition (100%) at 25% concentration. The inhibition rates observed at lower concentrations were 93.80 per cent (20%), 71.98 per cent (15%), 50.91 per cent (10%), and 22.95 per cent (5%). The number of sclerotial bodies also decreased significantly with increasing concentrations of sesame cake, with the lowest count (15) recorded at 15% per cent concentration (Table 1 and Fig. 3).
Other OSCs exhibited varying levels of antifungal efficacy. Castor and neem cakes displayed no inhibition at all tested concentrations, while groundnut cake exhibited minimal inhibition (9.56%) at 20 per cent concentration. Mustard cake showed modest inhibitory effects, with the highest inhibition (28.95%) observed at 25 per cent concentration. The mixture cake demonstrated concentration-dependent inhibition, with the highest inhibition rate of 31.72 per cent observed at 25%per cent concentration. In terms of sclerotial body production, the groundnut cake exhibited the highest sclerotial count (98) at 5 per cent concentration, while sesame cake resulted in the lowest count (15) at 15 per cent concentration. The results of the present study are consistent with those reported by Senjaliya and Nathawat (2015), who evaluated several OSCs, including mustard, groundnut, neem and castor cakes, for their efficacy against S. rolfsii. Their findings indicated that all OSCs, except FYM,
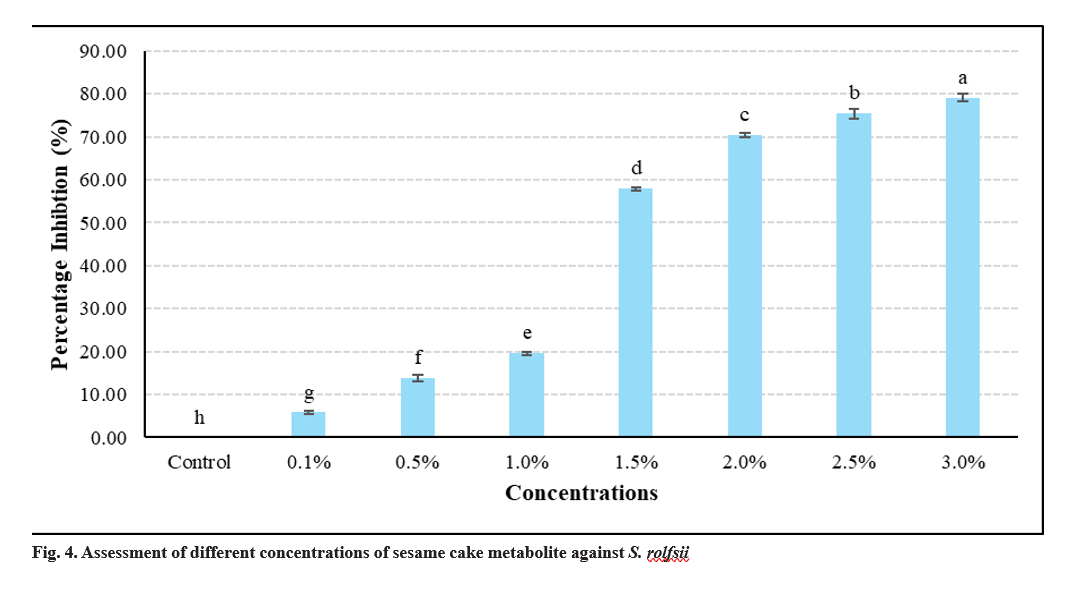
Table 2. Compounds with their retention time identified in GC-MS analysis of n-hexane fraction of sesame cake
significantly inhibited fungal growth, corroborating the results of the present study, where sesame cake and other OSCs showed substantial inhibitory effects. Similar inhibitory potential of OSCs against S. rolfsii has been reported by Anitha et al. (2019), who observed
80.11 per cent inhibition using mahua oil cake. The reduced efficacy of OSCs at lower concentrations may be attributed to differences in the bioactive compound concentrations and their thermostability, as suggested by You et al. (2019).
i. Bioactive compounds extracted from sesame cake aqueous extract
The antifungal potential of bioactive compounds extracted from sesame cake was evaluated using GC-MS analysis. The antifungal efficacy of the metabolite was tested at varying concentrations (0.1%, 0.5%, 1.0%, 1.5%, 2.0%, 2.5%, and 3.0%), by poisoned food technique. Mycelial growth was significantly inhibited in a concentration-dependent manner (p < 0.05). The highest concentration (3.0%) achieved 79.17% inhibition, while lower concentrations resulted in moderate inhibition rates of 70.42 per cent (2.0%), 75.42 per cent (2.5%), and 57.92 per cent (1.5%) (Fig. 4).
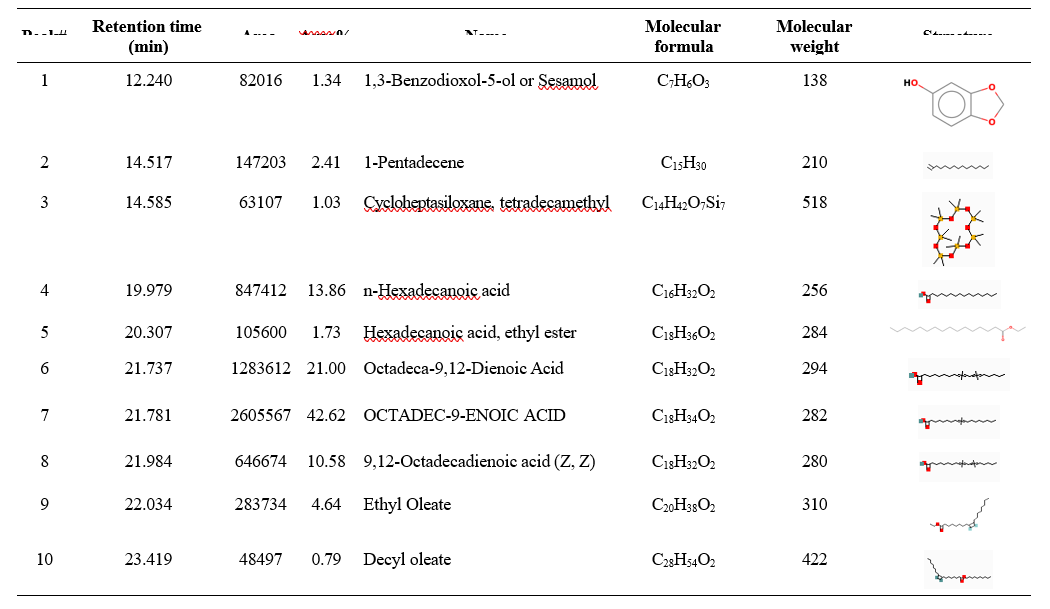
GC-MS analysis revealed that the primary bioactive compounds responsible for the antifungal activity of the sesame cake extract included oleic acid (42.62%), octadeca-9,12-dienoic acid methyl ester (21.00%), and 9,12-octadecadienoic acid (10.58%). These fatty acids have well-documented antifungal properties, suggesting their role as key bioactive agents. Additional compounds, such as n-hexadecanoic acid (13.86%) and sesamol (1.34%), also contributed to antifungal activity. The presence of ethyl oleate (4.64%) and hexadecanoic acid ethyl ester (1.73%) may enhance the solubility and availability of active compounds, thereby increasing antifungal efficacy (Table 2 and Fig. 5). These results are consistent with those of Ali et al. (2017), who observed antifungal properties of 9-octadecenoic acid (Z)-, methyl ester in methanolic root extracts of Chenopodium album. Similarly, Jangir et al. (2020) identified oleic and linoleic acids as predominant bioactive compounds in mustard OSCs. Ayyandurai et al. (2022) further demonstrated that n-hexadecanoic acid, linoleic acid, and oleic acid present in mahua oil cake significantly inhibited S. rolfsii at 5% and 10% concentrations. Collectively, these findings highlight the potential of fatty acids as antifungal agents in OSCs, further emphasizing the role of sesame cake as
a promising biocontrol strategy for managing S. rolfsii in groundnut.
Oilseed cakes (OSCs) have demonstrated significant potential as effective soil amendments for managing S. rolfsii in groundnut. Among the tested OSCs, sesame cake exhibited the highest antifungal activity, achieving complete inhibition of fungal growth and sclerotial germination at higher concentrations. The bioactive compounds present in sesame cake, such as oleic acid, linoleic acid derivatives, palmitic acid, and sesamol, play a crucial role in suppressing fungal growth. The superior performance of sesame cake compared to other OSCs highlights its potential as a sustainable and eco-friendly alternative to chemical fungicides. The findings emphasize the value of integrating sesame cake as a core component in the integrated management of S. rolfsii in groundnut cultivation.
LITERATURE CITED
Adiver, S. S. 2003. Influence of organic amendments and biological components on stem rot of groundnut, ISOR, National Seminar: Stress Management in Oil Seeds, January 28-30, 15-17.
Ali, A., Javaid, A and Shoaib, A. 2017. GC-MS analysis and antifungal activity of methanolic root extract of Chenopodium album against Sclerotium rolfsii. Planta Daninha. 35: 017164713.
Anitha, K., Thiruvudainambi, S., Harish, S and Kumar, A. 2019. Bio-efficacy of organic amendments and plant oils against Sclerotium rolfsii Sacc.: The incitant of Southern blight of tomato. Journal of Pharmacognosy and Phytochemistry. 8(3): 2202- 2205.
Ayyandurai, M., Akila, R., Mini, M. L and Manonmani, 2022.Gas chromatography–mass spectrometry (GC-MS) analysis of antimicrobial compounds produced by mahua oil cake against the stem rot pathogen-Sclerotium rolfsii. Journal of Applied and Natural Science.14(2): 600-606.
Bera, S.K., Kasundra, S.V., Kamdar, J.H., BC, A., Lal, C., Thirumalasmy, P.P., Dash, P and Maurya, A.K. 2014. Variable response of interspecific breeding lines of groundnut to Sclerotium rolfsii infection under field and laboratory conditions. Electronic Journal of Plant Breeding. 5(1): 22-29.
Blum, L.E.B and Rodriguez-Kabana, R. 2004. Effect of soil organic amendments on sclerotial germination, mycelial growth and Sclerotium rolfsii induced diseases. Fitopatalogia Brasileria. 29: 66-74.
Bosamia, T.C., Dodia, S.M., Mishra, G.P., Ahmad, S., Joshi, B., Thirumalaisamy, P.P., Kumar, N., Rathnakumar, A.L., Sangh, C., Kumar, A and Thankappan, R. 2020. Unraveling the mechanisms of resistance to Sclerotium rolfsii in peanut (Arachis hypogaea L.) using comparative RNA-Seq analysis of resistant and susceptible genotypes. PLoS One. 15(8): 0236823.
Dubey, S.C. 2002. Efficacy of some oil cakes and plant extracts against web blight of urd and mung bean caused by Thanatephorus cucumeris. Journal of Mycology and Plant Pathology. 32(2):158-161.
FAOSTAT, 2021. http://www.fao.org/faostat/en/#data/ QCAnnual report, 2024. 8-8-25.cdr
Jangir, M., Sharma, S and Sharma, S. 2020. Synergistic effect of oilseed cake and biocontrol agent in the suppression of Fusarium wilt in Solanum lycopersicum. Brazilian Journal of Microbiology. 51(4): 1929-1939.
Mc Clintock, J. A. 1917. The stem rot of peanut caused by Sclerotium rolfsii. Phytopathology. 7: 392-393.
Mehan, V.K and McDonald, D. 1990. Some important diseases of groundnut sources of resistance and their utilization in crop improvement. Country Training Course on Legumes Production. 9-17.
Mehan, V.K., Mayee, C.D., Mc Donald, D., Ramakrishna, N and Jayanthi, S. 1995. Resistance in groundnut to Sclerotium rolfsii-caused stem and pod rot. International journal of pest Management. 41(2): 79-83.
Pujer, S., Motagi, B.N., Kenchanagoudar, P.V and Gowda, M.V.C. 2021. Evaluation of Groundnut Segregating Population for Resistance to Sclerotium rolfsii. using an Efficient Field Screening Technique. Legume Research-An International Journal. 44(11): 1379-1385.
Senjaliya, B.D and Nathawat, B. D. S. 2015. Evaluation of bioagents, phyto-extracts and organic amendment on stem rot of groundnut. Annals of Plant Protection Sciences. 23(1): 98-101.
Sivaprakasam, K. 1991. Soil amendment for crop disease management. In P. Vidyasekaran (ed.) – Basic Research for crop disease management. Publishing house, Delhi. 306-315.
Thakkar, R.V., Chaudhary, S.M and Meena, R.L. 2018. Effect of organic amendments against stem rot of cluster bean caused by Sclerotium rolfsii. Int J Agric Sci. 10(6): 5628-5629.
Thangaraj, P., Subbiah, K.A., Sevugapperumal, N., Uthandi, S., Damodarasamy, A and Shanmugam,
2023. Activity of volatiles induced by microbes and natural plants stifled the growth of Pythium aphanidermatum-the damping off in Tomato. BMC plant biology. 23(1): 384.
USDA, 2023. https://ipad.fas.usda.gov/countrysummary/ Default.aspx?id=CH&crop=Peanut
You, X., Wakana, D., Ishikawa, K., Hosoe, T and Tojo, M. 2019. Antifungal Activity of Compounds isolated from bamboo vermicompost against Rhizoctonia solani AG1-IB. Advances in Microbiology. 9: 957-970.
- In Vitro Evaluation of Oil Seed Cakes (Oscs) as Sustainable Soil Amendments for the Management Sclerotium Rolfsii Pathogen in Groundnut
- Effect of Sowing Window on Nodulation, Yield and Post – Harvest Soil Nutrient Status Under Varied Crop Geometries in Short Duration Pigeonpea (Cajanus Cajan L.)
- Nanotechnology and Its Role in Seed Technology
- Challenges Faced by Agri Startups in Andhra Pradesh
- Constraints of Chcs as Perceived by Farmers in Kurnool District of Andhra Pradesh
- Growth, Yield Attributes and Yield of Fingermillet (Eleusine Coracana L. Gaertn.) as Influenced by Different Levels of Fertilizers and Liquid Biofertilizers

